
by dsaadmin | Medical Devices
Currently, disinfectants and sterilants that are intended for use on medical devices are regulated as drugs under the Food and Drug Regulations. However, Health Canada has communicated that they intend to reclassify these products as medical devices which means that...

by dsaadmin | Medical Devices
ISO will withdraw ISO 13485:2003 on March 1, 2019, three years after the publication of ISO 13485:2016. All Canadian and Foreign manufacturers of class II, III, and IV medical devices holding licences or applying for new or amended licences must complete the...

by dsaadmin | Medical Devices
As of July 16, 2016, decorative contact lenses will be regulated as Class II medical devices in Canada. Manufacturers will be required to obtain a medical device licence before advertising or selling decorative contact lenses. Health Canada will be...

by dsaadmin | Uncategorized
The Access to Information (ATI) Act allows Canadians to request copies of files which are within the government’s domain. ATI requests are often used within our industry to request copies of Product Registration Dossiers and even Health Canada Inspection...

by dsaadmin | Uncategorized
DSA is frequently approached by companies who want to seek us out as potential advertisers. Most are surprised to learn that DSA does not engage in traditional advertising models. We don’t go looking for clients; they find us. While clients find us in a...
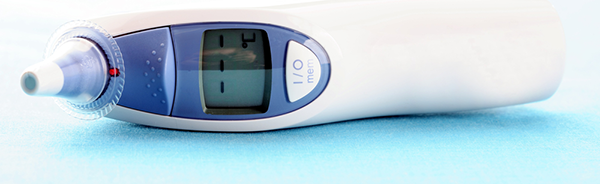
by dsaadmin | Medical Devices
Single-use medical devices are those labelled by their manufacturers to be used only once. For many years hospitals have been reprocessing some medical devices originally labelled for single use for cost savings. Reprocessing includes cleaning, sterilising and...

by dsaadmin | Announcement, Cosmetics
In the December 2015 update to the Cosmetic Ingredient Hotlist, a restriction was added to the Methylisothiazolinone/Methylchloroisothiazolinone (MI/MCI) entry. MI/MCI when used in combination is a prohibited substance in leave-on cosmetic products. An implementation...











